Hi, I am Alina Lösing, a bachelor’s student from Germany with the great opportunity to join the Cnidaria and Ctenophora team from the University Museum of Bergen for a 6 weeks Internship. This experience was not only a chance to further strengthen my passion in marine biology but also very helpful to gain valuable insight into research and museum operations.
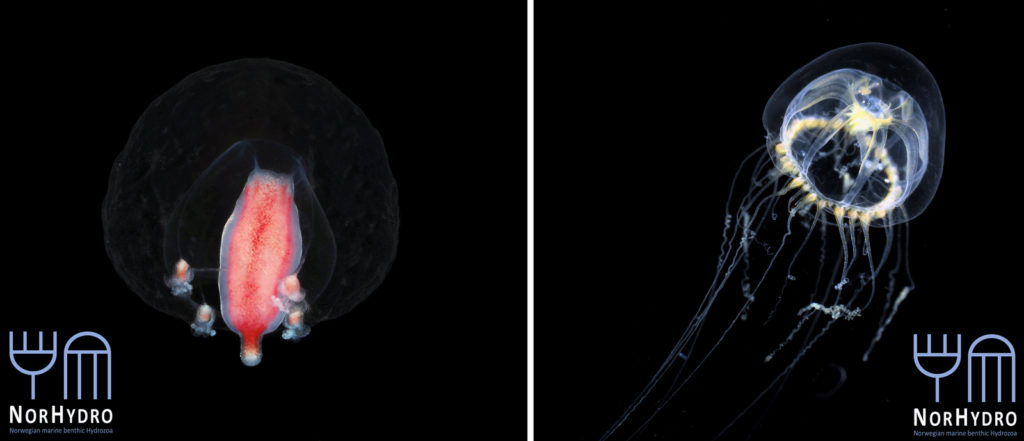
During my time at the museum, I had the chance to join various projects of Joan J. Soto-Angel, including POLE2POLE (Horizon 2020, MSCA) on bipolarity, and Artsprosjektet NoAH (Artsdatabanken) on Norwegian Arctic Hydrozoa. I even had the honor to contribute a small part to it! I focused mostly on the polyp of Stegopoma plicatile, a supposedly widely distributed species present also in both Arctic and the Antarctic waters.

Figure 1: Living specimen of Stegopoma plicatile collected during the OceanX cruise at 240 m depth (Pictures: Joan J. Soto-Angel)
The first step was to take pictures of the different specimens that were sampled and categorized as Stegopoma plicatile. Further I took pictures of many other species of Arctic hydrozoan polyps and hydromedusae. I learned some useful tricks with the camera while photographing the different species of polyps and medusae.
Part of the project is developing a reliable workflow which allows for a morphological distinction between polyps belonging to potentially different species previously defined molecularly from different areas worldwide. Therefore, I spent a lot of time working with the microscope collecting data which can be crucial to refining the morphological differences between different species. I measured length and width of 380 nematocysts!

Figure 2: Taking pictures of the samples with the microscope and the camera (Pictures: Joan J. Soto-Angel)
Furthermore, I underwent training in tissue sampling, a crucial step in preparing the samples for DNA sequencing. It’s all about preventing tissue contamination! Moreover, I had the opportunity to visit the DNA Lab, where I received instruction in their specialized techniques for DNA extraction, PCR, and electrophoresis. Many thanks Cessa for your time and dedication!
The highlights of my internship were of course both sampling trips I got to attend on board the Research Vessel Hans Brattstrøm. On the ship I learned a lot about the specific sampling methods for these incredibly delicate invertebrates. Despite experiencing a bit of seasickness the first time, I assisted with net deployment, sorting the animals, and collecting water samples from different depths.

Figure 3: Collecting water samples from a CTD; The Team during a successful sampling day (Pictures: Luis Martell, Praveen Raj)
Apart from an amazing Jellyfish Team, Bergen has many more attractions to offer. I highly recommend packing your hiking boots for your next visit, as the surrounding mountains offer breathtaking scenery. And if you share my passion for the ocean, a visit to the Nordnes Sjøbad is a must. Here you can take a refreshing dip in the frosty ocean before warming up in the heated saltwater pool.
I would like to take a moment to acknowledge Joan’s invaluable contribution to my internship at the University Museum of Bergen. Joan’s dedication and extensive knowledge have made my experience truly unforgettable.
-Alina