Special 20th anniversary 22.11.18
The Malacological Society of London
Conference talk about citizen scientists
A few weeks ago, Manuel Malaquias, Justine Siegwald and me travelled to London in order to attend the 20th anniversary conference of the Malacological Society of London, UK. This society is dedicated to research and education on molluscs. Although based in London (as the name refers to), the society is internationally orientated and welcomes all members interested in the scientific study of molluscs. The society was founded in 1893 and registered as a charity. One of the many activities of the society is to organize meetings and symposia, and this year it turned out to be a 20th anniversary of the molluscan forum!
It was an incredible interesting day with a lot of inspirational posters and talks. My mission for that day was to present our ‘Sea slugs of Southern Norway’ project with the emphasis on how citizen scientist made this project a success. I wanted to share with the audience how citizen scientists with the right approach could be the future for many scientific studies.
But first let us have a look into the meaning of citizen science. According to the Oxford English Dictionary, citizen science is scientific work undertaken by members of the general public, often in collaboration with or under the direction of professional scientists and scientific institutions. The term was first coined during the nineties in the United States of America. Since than it has grown in popularity, with multiple projects in the world that rely on the input of data generated by the general public. Some big and well-known examples are eBird, with roughly 411K users, Nasa Globe with 640K users, iNaturalist, with almost 1 million users and OPAL with 930K users and counting.
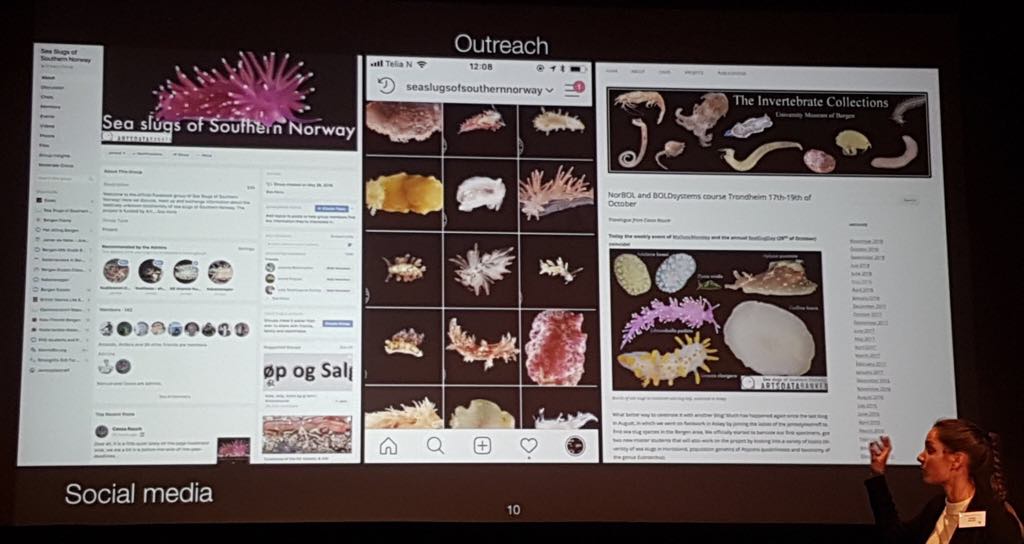
Since the beginning of this year we put a lot of effort in setting up a network of volunteers and underwater photographers. We got many good people willing to contribute to our project and most them are located in the South of Norway, but we also have a few located more further up North in Norway. Currently we have around 150 members directly and indirectly involved in helping us with our project. We try to involve our citizen scientist in the project as much as possible and one way is by reaching out to them via several social media platforms. For example, in our Facebook group community members can participate in discussions about species descriptions and share their findings etc. But we also have an Instagram account that functions as a pocket field guide for followers. Besides we try to keep everyone up to date about the project by regular posting blogs.
But our key element in this project definitely goes to the assembly and design of our sampling kits. These were designed specially for our citizen scientists in order to make collecting easier, more accessible and more standardized for everyone. By trying to standardize the collecting steps with so-called instructed sampling kits we minimized the errors that could occur during sampling of the data. The sampling kits contain plastic jars for the samples, fixative, preprinted labels and a USB flash drive with enough space for high-resolution pictures and a preset excel file that only needs filling in.

Example of the content of a citizen science sampling kit designed for the Sea slugs of Southern Norway project
We noticed that this approach worked out and the data quantity and quality increased as well the recruitment became easier. When we look at all the Norwegian sea slug records from the museum collection since the 19th century, it consists of roughly 1400 records. In just over six months time we see that the contribution of the citizen scientists covers almost half of that.
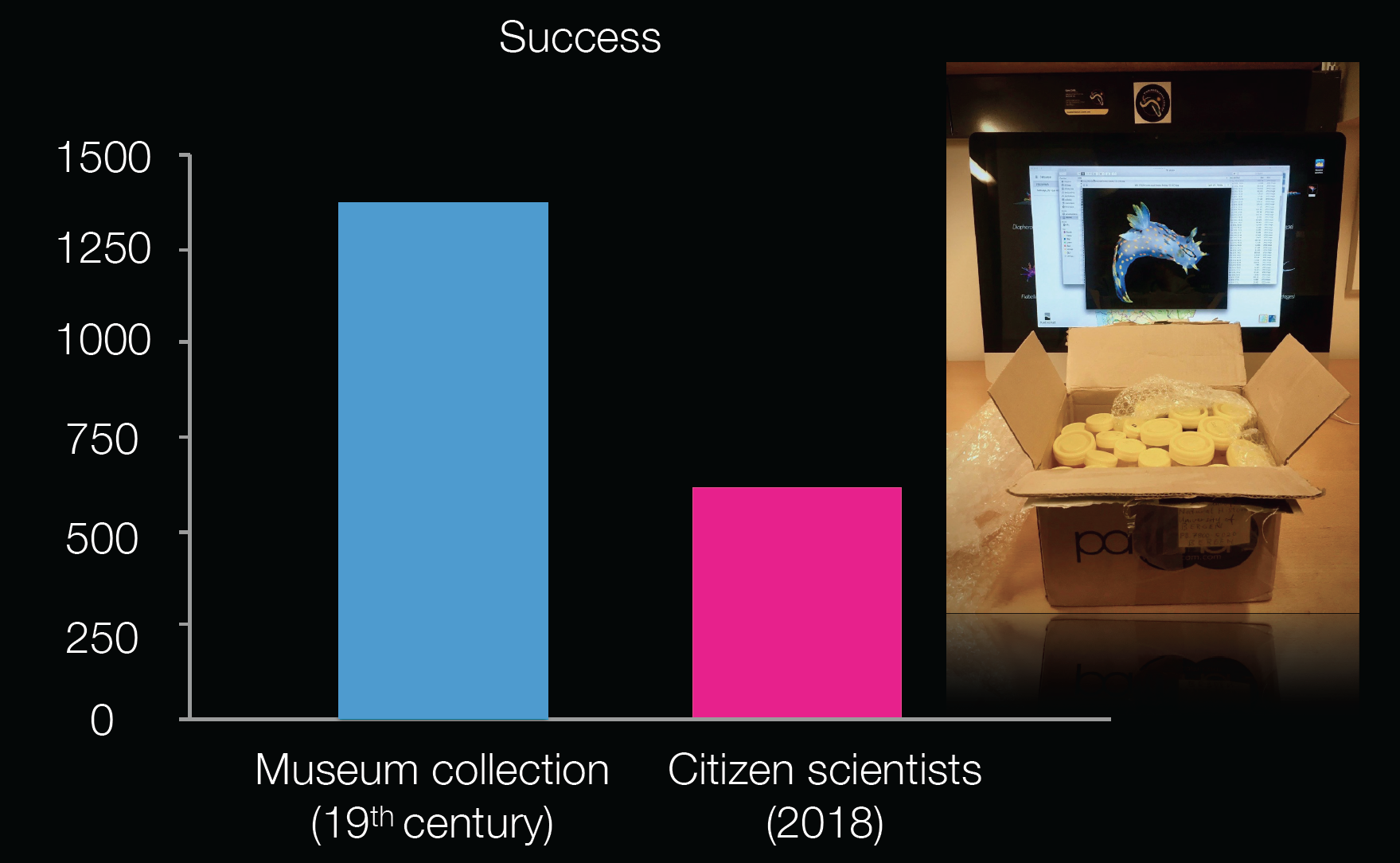
A comparison of the amount of records collected by citizen scientists since this year compared to our museum collection
The material that is sent in by the citizen scientists is at the moment being studied by us. We have two master students who will start working in January on a variety of taxonomic challenges by studying the different geographical material.
An example of this is Eubranchus farrani species complexes that have different color morphotypes from different geographical locations. Do we deal here with one species or multiple?
Stay tuned for a follow up!
Furthermore
Sea slugs of Southern Norway recently got its own Instagram account! Perfect for on the go if you would like to quickly check some species, click here https://www.instagram.com/seaslugsofsouthernnorway/ and don’t forget to follow us.
Become a member of the sea slugs of southern Norway facebook group, stay updated and join the discussion; https://www.facebook.com/groups/seaslugsofsouthernnorway/
Explore the world, read the invertebrate blogs!
-Cessa